Chemical Engineering Thermodynamics - Section 1 (3)
- Home
- Chemical Engineering
- Chemical Engineering Questions and Answers
- Chemical Engineering Thermodynamics - Section 1
Chemical Engineering Thermodynamics - Section 1
Fuels and Combustion is a field and there are problems available to
solve in this category. You will find the MCQs and can solve them easily.
| 17. | Boyle's law for gases states that |
|||||||
Answer: Option B Explanation: No answer description available for this question. Let us discuss. |
| 18. | Joule-Thomson experiment is |
|||||||
Answer: Option D Explanation: No answer description available for this question. Let us discuss. |
| 19. |
|
|||||||
Answer: Option D Explanation: No answer description available for this question. Let us discuss. |
| 20. | Linde gas liquefaction process employs cooling |
|||||||
Answer: Option A Explanation: No answer description available for this question. Let us discuss. |
| 21. | "Law of corresponding states" says that |
|||||||
Answer: Option A Explanation: No answer description available for this question. Let us discuss. |
| 22. | Degress of freedom at triple point will be |
|||||||
Answer: Option A Explanation: No answer description available for this question. Let us discuss. |
| 23. |
|
|||||||
Answer: Option B Explanation: No answer description available for this question. Let us discuss. |
| 24. | During Joule-Thomson expansion of gases |
|||||||
Answer: Option A Explanation: No answer description available for this question. Let us discuss. |

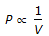 when temperature is constant.
when temperature is constant. = constant, for any gas.
= constant, for any gas. PCl3 + Cl2.Degree of dissociation of PCl5 will
PCl3 + Cl2.Degree of dissociation of PCl5 will