Chemical Engineering Thermodynamics - Section 1 (2)
- Home
- Chemical Engineering
- Chemical Engineering Questions and Answers
- Chemical Engineering Thermodynamics - Section 1
Chemical Engineering Thermodynamics - Section 1
Fuels and Combustion is a field and there are problems available to
solve in this category. You will find the MCQs and can solve them easily.
| 9. | ||||||||
Answer: Option A Explanation: No answer description available for this question. Let us discuss. |
| 10. | Equilibrium constant decreases as the temperature |
|||||||
Answer: Option A Explanation: No answer description available for this question. Let us discuss. |
| 11. | Which of the following is a thermodynamic property of a system ? |
|||||||
Answer: Option D Explanation: No answer description available for this question. Let us discuss. |
| 12. | In any spontaneous process, |
|||||||
Answer: Option C Explanation: No answer description available for this question. Let us discuss. |
| 13. | Pick out the correct statement: |
|||||||
Answer: Option B Explanation: No answer description available for this question. Let us discuss. |
| 14. | Out of the following refrigeration cycles, which one has maximum COP ? |
|||||||
Answer: Option B Explanation: No answer description available for this question. Let us discuss. |
| 15. | In a reversible chemical reaction (where, Δx = number of moles of products-number of moles of reactants ) |
|||||||
Answer: Option D Explanation: No answer description available for this question. Let us discuss. |
| 16. | 1st law of thermodynamics is nothing but the law of conservation of |
|||||||
Answer: Option C Explanation: No answer description available for this question. Let us discuss. |

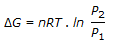 , gives the free energy change
, gives the free energy change